Hey readers! 👋
What a week it's been in the diabetes tech world! Barcelona hosted ATTD 2026, and the announcements kept rolling in. The big theme? Automation is getting smarter, and the line between "hybrid" and "fully" closed-loop systems is finally starting to blur. Plus, we've got some fascinating cell therapy updates that had me doing a double-take at the data. Let's dive in!
🔬 This Week's Highlights
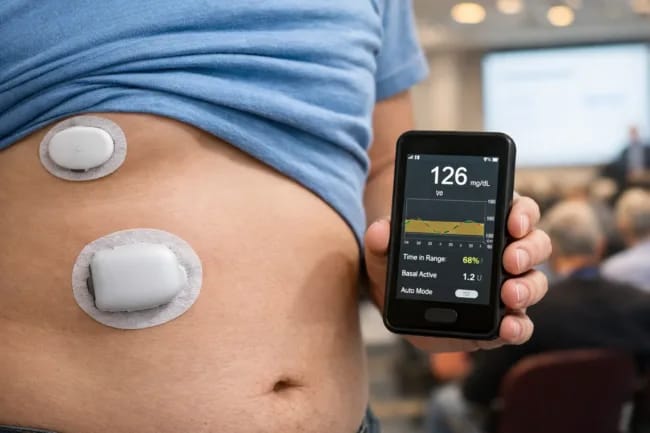
Insulet's EVOLUTION 2 Study Shows Promising Results for Fully Closed-Loop System - While this study focused on Type 2 diabetes, the implications for T1D are worth watching. The fully closed-loop system achieved 68% time-in-range without any mealtime boluses, a 24% improvement over standard injection therapy. No severe hypoglycemia or DKA events were reported across the 24-participant trial. – MedTechDive
"Developing and bringing to market a fully closed-loop AID system that delivers therapy effortlessly and adapts automatically with no mealtime interactions or adjustments is more than evolutionary; it's revolutionary," said Dr. Trang Ly, Insulet's Chief Medical Officer.
Insulet plans a pivotal trial this year, with a 510(k) filing targeted for 2027 and commercial launch in 2028. The technology could eventually expand to T1D applications, so this is definitely one to keep on your radar.
Sana Biotechnology Reports 14-Month Success with Immunosuppression-Free Islet Cell Transplant - Here's something genuinely exciting: Sana's UP421 therapy, using hypoimmune-modified islet cells, has shown sustained beta-cell survival and function for 14 months in a T1D patient without immunosuppression. C-peptide levels at month 14 were comparable to the first six months, and interestingly, tighter glycemic control between months 12-14 correlated with improved insulin secretion. – Wallstreet Online
The company plans to file an IND for its stem-cell-derived therapy SC451 and launch a Phase 1 trial later this year. No safety issues were identified, which is encouraging for this early-stage work.
🎯 ATTD 2026 Conference Updates
Breakthrough T1D Highlights Key Developments at ATTD - The Barcelona conference showcased several important developments. Global screening programs are expanding, with pilot programs demonstrating how early autoantibody detection can prevent DKA and enable timely enrollment in disease-modifying trials. Teplizumab (Tzield) implementation was a major topic, with discussions on the multidisciplinary support needed for infusion, monitoring, and patient education. – Breakthrough T1D
"With proper monitoring, DKA is almost completely preventable."
Emerging therapies discussed included AI-based closed-loop delivery systems, gene-edited muscle-cell insulin production from Kriya Therapeutics, and scalable manufactured islet cell transplants. The conference emphasized that translating laboratory advances into everyday patient care requires robust clinical infrastructure and training for new endocrinologists.
📱 Tech & Device News
The twiist Insulin Pump Launches Nationwide in the U.S. - Sequel Med Tech has expanded its twiist AID system from limited release to full nationwide availability. The pump uses the Tidepool Loop algorithm and features iiSure Technology, which measures insulin volume with every microdose and can detect delivery issues up to 9x faster than other systems. New integrations include the Eversense 365 implantable CGM, with plans to add the DBLG2 algorithm from Diabeloop in 2027. – Diabetech
SiBionics Showcases AI-Enhanced CGM Features at ATTD - SiBionics unveiled AI-driven features for their ultra-compact GS3 CGM platform, including multi-day time-in-range overviews, spike analysis, smart voice logging, and meal-impact insights. The company is also partnering with PharmaSens to develop an all-in-one insulin patch pump. – Drug Delivery Business
Afon Technology Presents Non-Invasive CGM Data - Afon's Glucowear needle-free monitor achieved roughly 75% overall accuracy in distinguishing normal from elevated glucose levels in its first trial, with 87% sensitivity and 85% specificity. The company will present findings at ATTD and initially targets non-insulin-dependent T2D before expanding to broader populations. – Drug Delivery Business
📋 Quick Hits
Mayo Clinic Lists 68 Active T1D Clinical Trials - Studies range from new insulin formulations to hybrid closed-loop systems, biomarkers, genetics, and behavioral interventions. – Mayo Clinic
Juicebox Podcast Discusses Cost-Saving Tips - Host Scott Benner shares how switching supplies from DME to pharmacy co-pays can save up to $2,000 annually, plus a listener's experience with diabetic retinopathy improvement after better glucose control. – Scott Benner
Dexcom G7 Offers Predictive Low Alerts - The system's "Urgent Low Soon" feature can notify users up to 20 minutes before a hypoglycemic event, with integration for Apple Watch and smartphone apps. – Dexcom
🛠️ Tools Worth Knowing
mySugr App Consolidates Diabetes Data - This app, developed by people with diabetes, brings together blood sugar readings, meals, activity, and insulin doses into one platform. Features include a Bolus Calculator, estimated HbA1c, meal photo logging, and pattern visualization over 7, 14, 30, or 90 days. Basic features are free with Accu-Chek device integration. – mySugr
That's a wrap for this week! The pace of innovation continues to accelerate, and it's encouraging to see so many different approaches being pursued simultaneously. Whether it's fully automated insulin delivery, cell therapies without immunosuppression, or AI-enhanced glucose monitoring, the future of T1D management keeps getting more interesting.
Made with ❤️ by Data Drift Press
Hit reply with your questions, comments, or feedback - I'd love to hear from you!